Nuclear Medicine
Research Focus
General Facts
Research
Selected Publications
Selected Funding, Collaboration
Devices & Services
Keywords: Peptide receptor radionuclide therapy, peptide ligand radionuclide therapy, radioiodine refractory thyroid cancer, hormone refractory prostate cancer, neuroendocrine tumours, thera(g)nostics
Research (ÖSTAT Classification) : 302054, 301904, 301206
Research Focus
The Department of Nuclear Medicine is best known for its work with radiolabelled peptides, both for diagnostic and for therapeutic purposes, a theme that we have systematically explored over the last three decades. We are developing a variety of radiopharmaceuticals for different targets for clinical use. Our goal is to engineer more effective ligands/peptides/antibodies "thera(g)nostics" for individualised treatment, mainly in the area of oncology.
General Facts
The Department of Nuclear Medicine accelerates the translation of preclinical radiopharmaceutical research development (focus on radiolabelled peptides) into clinical applications, towards imaging of biomarkers used for cancer treatment (70% of clinical routine), treatment of neurological impairment (20% of clinical routine) and cardiac disease (10% of clinical routine). The structure of the Department of Nuclear Medicine is based on a very creative, productive, well-funded and internationally respected, high-quality, preclinical research and development unit. This group consists of several radiochemists/pharmacists, medical physicists and PhD-students. Their work results in the construction of radiotracers, which use different modal systems including a variety of radiolabelled peptide analogues such as for somatostatin, vasoactive intestinal peptide (VIP), cholecystokinin (CCK-2/gastrin) and prostate-specific membrane antigen (PSMA) ligand for specific tumour targeting. Other important developments are based on Arg-Gly-Asp (RGD) for imaging of angiogenesis in tumour lesions and on hepatic binding protein imaging with galactosylated albumin (NGA) for functional liver reserve estimation. Radiopharmaceuticals are produced at clinical grade in our dedicated laboratories, for use in SPECT/CT or PET/CT studies. About 30 whole-body PET/CT studies are performed daily in our PET centre. Patients assessed by SPECT/CT dosimetry studies are treated in our Nuclear Medicine Therapy Unit with high-dose thera(g)nostics. Radioiodine ablation therapy of thyroid cancer remnants, peptide receptor radionuclide therapy (PRRT) of neuroendocrine tumour patients and peptide ligand radionuclide therapy (PLRT) of prostate cancer patients are currently our most important therapy tools. The Department of Nuclear Medicine is involved not only in many clinical studies with radiolabelled thera(g)nostics, particularly industry-sponsored Phase I/IIa trials, but also in academic Phase I trials with radiopharmaceuticals developed in-house.
Research
The research activities are focused on preclinical research dedicated to the optimisation and improvement of radiolabelling procedures for established radiopharmaceuticals, the in-house preparation of new radiopharmaceuticals for clinical studies and the preclinical development of new radioligands for molecular imaging and therapeutic purposes. Different research projects illustrate the activities in this field. Some of the thera(g)nostics developed in the Department of Nuclear Medicine have already entered larger clinical Phase I/II trials.
Siderophores for molecular imaging and thera(g)nostics
Leads: Clemens Decristoforo and Bernhard Nilica
Nuclear medicine can play a vital role in the field of infection, through molecular imaging and thera(g)nostics. Siderophores are low-molecular mass, Fe3+-specific chelators secreted by bacteria and fungi. The use of siderophores labelled with gallium-68 for PET has been a focus of preclinical research for many years. In a current, preclinical project, Triacetylfusarinine C (TAFC) – the main extracellular siderophore of Aspergillus fumigatus (AFU), responsible for severe invasive fungal infections – is modified for novel applications. The main objective is to develop and characterise novel analogues of TAFC targeting AFU in vivo and to establish a novel concept for “thera(g)nostics” (combining diagnosis and therapy) of invasive aspergillosis. One example is the chemical modification of TAFC with fluorescent dyes (PhD thesis of Joachim Pfister). This allows so-called “hybrid imaging” by combining optical imaging (650 – 800 nm excitation wavelength) with PET. An example of microscopy of such a compound and in vivo imaging in a rat model infected with AFU in the lung by PET/CT and optical imaging is shown in Fig. 1 [1]. Therapeutic applications, modifying siderophores with antifungal molecules, are currently under investigation to produce thera(g)nostic compounds. This work is financed by FWF (P 30924‐B26) and supported by interdisciplinary (Hubertus Haas in the Molecular Biology division and Alexander Lichius in the Department of Microbiology) and international (Milos Petrik at the Institute of Molecular and Translational Medicine in the Czech Republic) collaborations. Other research topics cover the use of radiolabelled siderophores for bacterial infections, including a variety of preclinical approaches.
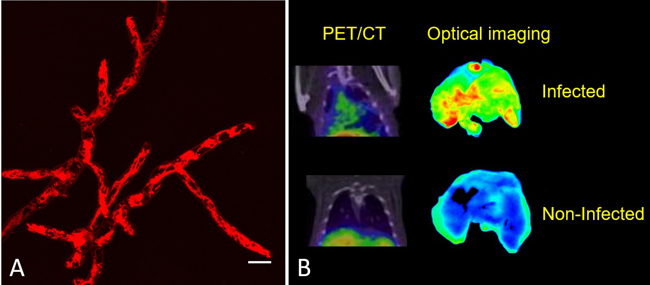
Fig. 1: (A) Live-cell image of AFU hyphae with [Fe]DAFC-Cy5 after 15 min, highlighting intracellular mitochondria. Scale bar: 10µm. (B) Left column: Coronal μPET/CT slices of AFU-infected (top) and non‐infected animals (bottom) 45 min p.i. in immunocompromised Lewis rats (approx. 5 10 MBq injected dose) Right column: Fluorescence images of corresponding lungs excised from infected and non‐infected rats 1.5 h after injection. Images reproduced from Pfister et al. [1].
Based on the successful preclinical development of 68Ga-labelled siderophores, translation into clinical practice has been initiated for PET infection imaging. A Phase I/IIa clinical trial using the clinically approved drug Deferoxamin (Desferal ®) labelled with Gallium-68 for imaging bacterial infections was submitted and approved by the ethics committee and regulatory authority (EudraCT no.: 2020-002868-31). This clinical trial, led by Bernhard Nilica and starting in 2021, is supported by FWF within the KLIF-track (Proj. no. KLI 909-B to C. Decristoforo).
Cholecystokinin Receptor Thera(g)nostics
Leads: Elisabeth von Guggenberg and Christian Uprimny
Radiolabelled minigastrin (MG) analogues with specific binding to the cholecystokinin-2 receptor (CCK2R) provide a very sensitive molecular imaging technique for the detection of medullary thyroid carcinoma (MTC) and other CCK2R-expressing malignancies, such as gastroenteropancreatic and bronchopulmonary NET and small-cell lung cancer. When radiolabelled with ß-emitting radionuclides, these analogues should become useful for targeted radiotherapy [2].
New radiolabelled peptide analogues are being developed to overcome high kidney uptake and low metabolic stability, which especially limit the therapeutic application of the currently available radiolabelled minigastrin analogues. By exploring novel stabilisation strategies within the receptor-binding motif of the linear amino acid sequence, it has been possible to develop novel minigastrin analogues with improved stability and retained receptor binding. Twelve different, new peptide analogues conjugated to the macrocyclic chelator DOTA for radiolabelling with different radioisotopes, such as indium-111 SPECT, gallium-68 for PET and lutetium-177 for therapeutic use, have been evaluated. The preclinical investigations covered the characterisation of stability in serum and tissue homogenates, the evaluation of CCK2R-binding and cell internalisation on CCK2R-expressing cell lines as well as biodistribution studies in a mouse tumour model that also included small-animal imaging and dosimetry studies. This work was financed by FWF project – P27844 and supported by international collaborations (Jane Sosabowski at the Centre for Molecular Oncology, Barts Cancer Institute, Queen Mary University of London and Peter Laverman in the Department of Nuclear Medicine in Nijmegen, Netherlands). It was possible to select one promising candidate for clinical translation from these studies [3]. A first proof of principle of high-sensitivity PET/CT imaging was achieved for 68Ga-DOTA-MGS5 in a 75-year-old female patient with recurrent MTC (Fig. 2) and graded as “image of the month” by the European Journal of Nuclear Medicine and Molecular Imaging [4]. Consequently, a prospective Phase l/lla study was designed and patients are currently being recruited (“Phase I/IIa study to evaluate the safety, tolerability, whole-body distribution and preliminary diagnostic performance of a novel 68Ga-labelled minigastrin analogue in patients with advanced neuroendocrine tumours” (EudraCT No. 2020-003932-26).
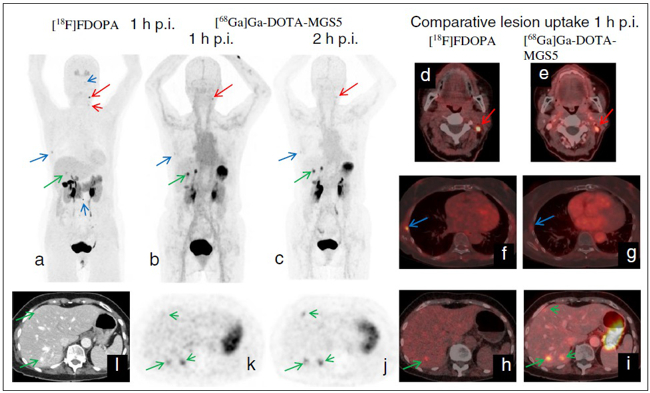
Fig. 2: PET CT imaging in a patient with recurrent MTC using [18F]FDOPA (a, d, f, h) and [68Ga]Ga-DOTA-MGS5 (b/c, e, g, i – l), showing several pathologic lesions suggestive of metastases (red arrows: lymph nodes; blue arrows: bone lesions; green arrows: liver lesions); three liver lesions were undetectable by standard imaging with [18F]F-DOPA.
Images reproduced from [4].
Angiogenesis Imaging – non-invasive determination of the integrin αvβ3 expression
Leads: Roland Haubner and Gianpaolo di Santo
Tumour-induced angiogenesis is a key process in the development of cancer. One receptor involved in this complex process is integrin αvβ3. We developed a 68Ga-labelled small cyclic peptide ([68Ga]NODAGA-RGD), which binds with high affinity to this receptor [5]. Based on the very promising preclinical results and on the initial data (Fig.3) of a Phase I study [6], a subsequent clinical study was initiated to investigate the potential of this radiopharmaceutical (EudraCT No 2018-001016-30): “Prospective study of 68Ga-NODAGA-RGD-PET for the non-invasive determination of the integrin αvβ3 expression in patients with mamma carcinoma, glioblastoma, renal cell cancer, non-small-cell lung cancer or neuroendocrine tumours.” (thesis of Stephan Zirknitzer).
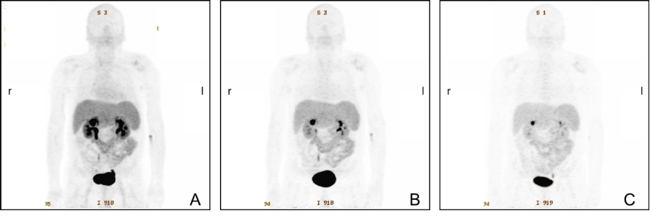
Fig. 3: Maximum-intensity projections from static [68Ga] NODAGA-RGD PET scans of a male patient with hepatocellularcarcinoma at 13 min (A), 40 min (B), and 76 min (C) after tracer injection. The tracer shows rapid predominant renal elimination with the highest activity in the bladder, kidneys, liver, spleen and intestine. Low background activity is found in the brain, thorax and extremities, resulting in a low radiation dose for the patient. For all three images, the grey-scale is set to the same value. Image from [6].
Non-Invasive determination of the functional liver reserve
Leads: Roland Haubner and Irene Virgolini
The asialoglycoprotein receptor (ASGR) is exclusively expressed on the basolateral side of hepatocytes and this makes it a promising target for drug delivery into hepatocytes. Moreover, because of its restricted expression, the ASGR is an optimal target structure for non-invasive monitoring of the functional liver reserve. Our initial developments resulted in 68Ga-NOTA-GSA, which demonstrated high metabolic stability and confirmed the superiority of PET for imaging the functional liver reserve [7] (Fig. 4). Unfortunately, the requirements for production of a GMP-compliant labelling precursor have increased in recent years. Consequently, despite the good imaging performance, translation of this human serum albumin-based labelling precursor into clinical practice has not been possible, which indicates the need for radiolabelled synthetic derivatives of low molecular weight as galactose carriers. To satisfy these requirements, the development of such derivatives has been started and a corresponding proposal for funding has been submitted to FWF (P 34802-B).
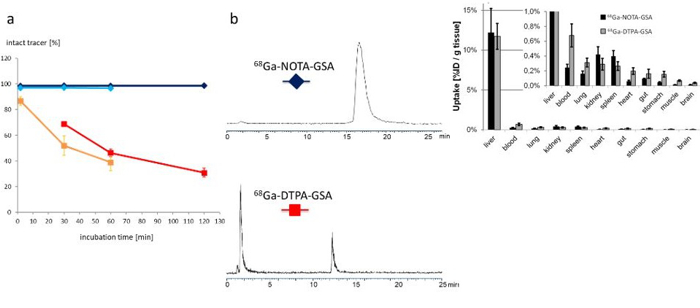
Fig. 4: Left side (a and b): Metabolic stability is clearly increased for the 68Ga-NOTA-GSA (blue lines) compared with the 68Ga-DTPA-GSA (red line) and the 99mTc-DTPA-GSA (orange line). Right side: The newly developed 68Ga-NOTA-GSA shows high activity accumulation in the target organ (liver) and only marginal activity concentration in other organs. Because of the metabolic stability, activity concentration in blood and most other organs is even lower than for the lead structure. Images reproduced from [7].
MITIGATE, NeoFind, Neother and NeoRay Clinical Studies
Leads: Bernhard Nilica, Clemens Decristoforo, Christine Rangger and Irene Virgolini
The MITIGATE study consisted of concerted action in which several potential imaging diagnostics were evaluated. In the WP7 package, the Department of Nuclear Medicine performed the first diagnostic in vivo studies in patients with GIST tumours (“A Phase I/IIa study to evaluate safety, biodistribution, dosimetry and preliminary diagnostic performance of 68Ga-NeoBOMB1 in patients with advanced TKI-treated GIST using positron-emission tomography/computer tomography (PET/CT).” EudraCT: 2016-002053-38, [8]. Consequently, the industry-sponsored (AAA/Novartis) NeoFIND study (“Phase II study of preliminary diagnostic performance of [68Ga]-NeoBOMB1 in adult patients with malignancies known to overexpress gastrin releasing peptide receptor” (EudraCT: 2017-003432-37) was implemented in Innsbruck and resulted in the NeoTher study “Phase Ia-Ib, safety, tolerability, whole-body distribution, radiation dosimetry of [177Lu]-NeoBOMB1 administered in subjects with gastrin-releasing peptide receptor (GRPR) positive relapsed or refractory metastatic breast and prostate cancer” and in the NeoRay study (“A Phase I/IIa open-label, multi-centre study to evaluate the safety, tolerability, whole-body distribution, radiation dosimetry and anti-tumour activity of [177Lu]-NeoB administered in patients with advanced solid tumors known to overexpress gastrin-releasing peptide receptor (GRPR).” (EudraCT: 2018-004727-37), which is currently recruiting tumour patients.
Prostate Cancer Thera(g)nostics
Leads: Christian Uprimny and Irene Virgolini
New receptor radiotracers binding to the prostate-specific membrane antigen (PSMA) – which is significantly increased in prostate cancer (PC) cells – have been proposed for PET imaging. The 68Ga-PSMA ligand HBED-CC has proven its feasibility for the detection of PC relapses and metastases with high sensitivity. In recent years, many clinical studies have demonstrated the great potential of 68Ga-PSMA PET/CT in patients with biochemical relapse [9]. Innsbruck was part of the multicentre academic trial that resulted in implementation of this new PET scan in guidelines for the work-up of PC patients: “An open-label, single-arm, rater-blinded, multicentre Phasel/ll study to assess safety and diagnostic accuracy and radiotherapeutic implications of pre-operative 68Ga-PSMA-11 PET/CT imaging in comparison with histopathology, in newly-diagnosed prostate cancer (PCA) patients at high risk of metastasis, scheduled for radical prostatectomy (RP) with extended pelvic lymph node dissection (EPLND) (EudraCT: 2016-001815-19).
Based on the high-level expression of PSMA in PC cells we have started to use high-dose 177Lu-PSMA ligand to treat patients who have metastasised disease. Results over recent years demonstrate high tumour control ability of this radiopharmaceutical with significant implications for future PC therapy protocols [10]. A WARMTH-sponsored retrospective multicentre trial [11] approved by Innsbruck ethics committee showed significant results that are superior to chemotherapy. Combination schedules and new radioligands as well as mixtures of radioligands for treatment of PC are being discussed. Industry-sponsored studies are currently in preparation such as the Blue Earth-sponsored study “An open-label, Phasel/ll dose-escalation, safety and proof-of-concept study of 177Lu-rhPSMA in men with castration resistant prostate cancer” and the NOVARTIS study “Phase lll, open-label, multi-centre, randomized study comparing 177Lu-PSMA-617 vs. a change of novel androgen axis drugs in the treatment of taxane naïve men with progressive metastatic castrate resistant prostate cancer”.
Somatostatin Receptor Thera(g)nostics
Leads: Margerida Rodrigues, Hanna Svyridenka and Irene Virgolini
Peptide receptor RT (PRRT) has been recognised as a promising treatment for neuroendocrine (NET) patients. The use of 18F-FDG PET in NET is still controversial, whereas 68Ga-labelled somatostatin analogues are recognised radiopharmaceuticals. The long-term survival and efficacy of a second PRRT course with somatostatin analogues 177Lu-DOTATE in patients with advanced gastroenteropancreatic NET was evaluated (thesis of Kevin Winkler). Furthermore, the value of 18F-FDG PET/CT in these patients was assessed. Forty patients with GEP NET, who underwent two PRRT courses with 177Lu-DOTATATE and combined examinations with 68Ga-DOTA-TOC and 18F-FDG PET/CT, were evaluated (Fig. 5). After the second PRRT course, 2 patients (5.0%) were in partial remission, 21 patients (52.5%) experienced stable disease and 17 patients (42.5%) had progressive disease. The median overall survival was 122 months. After the second PRRT course, the median overall survival was significantly higher (P=0.033) in the 18F-FDG-negative group compared with the 18F-FDG-positive group (145.50 versus 95.06 months, respectively). The median time to progression was 19.37 months. In conclusion, a second PRRT course with 177Lu-DOTATE is an effective treatment approach for GEP NET patients with disease progression. A change in 18F-FDG status after PRRT may predict disease course and survival. Patients who are 18F-FDG negative have significantly longer overall survival than those who are 18F-FDG positive [12].
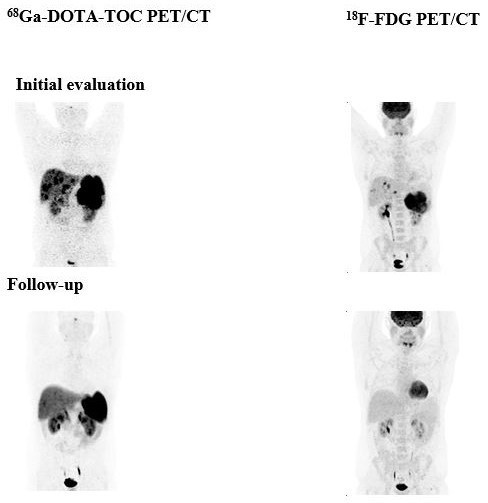
Fig. 5: A 30- year-old male patient with NET G2 in the pancreatic tail and Ki-67 12% in liver metastases. 68Ga-DOTA-TOC and to a lesser extent 18F-FDG PET show multiple liver metastases before PRRT and disease remission after 4 PRRT cycles with 177Lu-DOTA-TATE (cumulative dose of 22.97 GBq).
18F-FDG positivity provides the basis for a combination of PRRT with chemotherapy (in collaboration with the oncology department) and the first patients are being treated with this combination scheme in the Department of Nuclear Medicine (lead Hanna Svyridenka).
Over the last two decades, various somatostatin analogues have been implemented for thera(g)nostic use. The IPSEN-sponsored clinical studies using a somatostatin antagonist were:
“A multicentre, randomized, dose-confirmation, factorial Phase 2 study to evaluate the optimal dose of 68Ga-OPS202 as a PET imaging agent in subjects with gastroenteropancreatic neuroendocrine tumour (GEPNET)” EudraCT: 2016-004928-39.
68Ga-OPS202 in Breast Cancer Patients: “A non-randomized Phase ll study to evaluate the optimal uptake time of 68Ga-OPS202 as a sstr2 positive PET imaging agent in subjects with newly diagnosed breasts cancer.” EudraCT: 2018-000028-33/NCT.
“A multicentre, open-label Phase l/ll study to evaluate the safety, tolerability, biodistribution and anti-tumour activity of 177Lu-OPS201 with companion imaging 68Ga-OPS202 PET/CT in previously treated subjects with locally advanced or metastatic cancers expressing somatostatin receptor 2 (SSTR2)” EudraCT: 2017-005173-39/NCT.
The overexpression of somatostatin receptors (SSTR) predominantly of subtype – 2 (SSTR2) in neuroendocrine neoplasms is an established target for radiopharmaceuticals, which allows prediction and evaluation of response to various available therapies. The development of a receptor antagonist labelled with technetium-99m for SPECT with superior imaging properties is the aim of the ERA-PerMED project “TECANT” [https://www.era-learn.eu/network-information/networks/era-permed/1st-joint-transnational-call-for-proposals-2018/] supported by FWF (proj. no. I 4220-B). SSTR antagonists were prepared and compared preclinically in respect of their pharmacological properties in vitro and in vivo to provide a basis for clinical translation. [99mTc]-TECANT 1, which has higher tumour uptake and retention, was selected. Fig. 6 shows a micro-PET study in tumour-bearing mice displaying excellent targeting of [99mTc]-TECANT 1, from [13]. Radiation dose estimates predict a low acceptable radiation dose for patients. The project is a collaboration between universities in Krakow in Poland, Ljubljana in Slovenia and Basel in Switzerland. The clinical trial will begin in 2021.
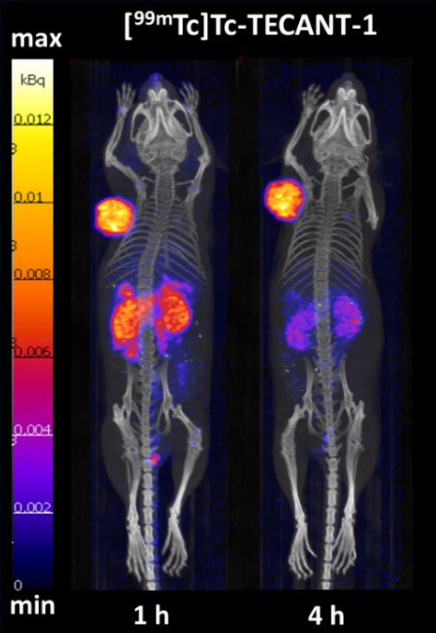
Fig. 6: SPECT/CT images of [99mTc]Tc-TECANT-1 in nude mice bearing HEK-SST2 (+) tumour xenografts (left shoulder) at 1 and 4 hours p.i., reproduced from [13]
Neurotensin Receptor Imaging
Leads: Irene Virgolini and Feng Wand, Nanjing, China
In collaboration with Professor Feng Wang from Nanjing in China (where Irene Virgolini is an appointed Visiting Professor), initial 68Ga-labelled-Neurotensin analogue- PET/CT scans were performed on patients with pancreatic cancer [14]. The positive scans point to a future role for such analogues and possibly to identification of a stable 177Lu-labelled neurotensin analogue for therapy.
Selected Publications
- Pfister, J.; Summer, D.; Petrik, M.; Khoylou, M.; Lichius, A.; Kaeopookum, P.; Kochinke, L.; Orasch, T.; Haas, H.; Decristoforo, C. Hybrid Imaging of Aspergillus fumigatus Pulmonary Infection with Fluorescent, 68Ga-Labelled Siderophores. Biomolecules 2020, 10, 168.
- Klingler M, Hörmann AA, Guggenberg EV. Cholecystokinin-2 Receptor Targeting with Radiolabeled Peptides: Current Status and Future Directions. Curr Med Chem 2020, 27, 7112-7132
- Klingler M, Summer D, Rangger C, Haubner R, Foster J, Sosabowski J, Virgolini I, Decristoforo C, von Guggenberg E. DOTA-MGS5, a new cholecystokinin-2 receptor targeting peptide analog with optimized targeting profile for theranostic use. J Nucl Med. 2019 Jul;60(7):1010-1016.
- Uprimny C, von Guggenberg E, Svirydenka A, Mikołajczak R, Hubalewska-Dydejczyk A, Virgolini IJ. Comparison of PET/CT imaging with [18F]FDOPA and cholecystokinin-2 receptor targeting [68Ga]Ga-DOTA-MGS5 in a patient with advanced medullary thyroid carcinoma. Eur J Nucl Med Mol Imaging 2020, Epub ahead of print.
- Knetsch PA, Petrik M, Griessinger CM, Rangger C, Fani M, Kesenheimer C, von Guggenberg E, Pichler BJ, Virgolini I, Decristoforo C, Haubner R. [68Ga]NODAGA-RGD for imaging αvβ3 integrin expression. Eur J Nucl Med Mol Imaging 2011, 38:1303-1312.
- Haubner R, Finkenstedt A, Stegmayr A, Rangger C, Decristoforo C, Zoller H, Virgolini IJ. [68Ga]NODAGA-RGD - Metabolic stability, biodistribution, and dosimetry data from patients with hepatocellular carcinoma and liver cirrhosis. Eur J Nucl Med Mol Imaging. 2016; 43:2005-2013.
- Haubner R, Schmid AM, Maurer A, Rangger C, Roig LG, Pichler BJ, Virgolini IJ. [68Ga]NOTA-Galactosyl Human Serum Albumin: a Tracer for Liver Function Imaging with Improved Stability. Mol Imaging Biol. 2017; Feb 13. doi: 10.1007/s11307-017-1046-1. [Epub ahead of print].
- Gruber L, Jiménez-Franco LD, Decristoforo C, Uprimny C, Glatting G, Hohenberger P, Schoenberg SO, Reindl W, Orlandi F, Mariani M, Jaschke W, Virgolini I. MITIGATE-NeoBOMB1, a Phase I/IIa Study to Evaluate Safety, Pharmacokinetics and Preliminary Imaging of 68Ga-NeoBOMB1, a Gastrin-Releasing Peptide Receptor Antagonist, in GIST Patients. J Nucl Med. 2020 Dec;61(12):1749-1755. pii: jnumed.119.238808. doi: 10.2967/jnumed.119.238808. Epub 2020 Apr 24.
- Uprimny C, Bayerschmidt S, Kroiss AS, Fritz J, Nilica B, Svirydenka A, Decristoforo C, Di Santo G, von Guggenberg E, Horninger W, Virgolini I. Impact of forced diuresis with furosemide and hydration on the halo artefact and intensity of tracer accumulation in the urinary bladder and kidneys on [68Ga]Ga-PSMA-11-PET/CT in the evaluation of prostate cancer patients. Eur J Nucl Med Mol Imaging. 2020 May 8. doi: 1.1007/s00259-020-04846-3
- Maffey-Steffan J, Scarpa L, Svirydenka A, Bernhard N, Mair C, Buxbaum S, Bektic J, vonGuggenberg E, Uprimny C, Horninger W, Virgolini I. The 68Ga/177Lu-theragnostic concept in PSMA-targeting of metastatic castration-resistant prostate cancer: impact of post-therapeutic whole-body scintigraphy in the follow-up. Eur J Nucl Med Mol Imaging. 2020 Mar;47(3):740.
- Ahmadzadehfar H, Rahbar K, Baum R, Seifert R, Kessel K, Bögemann M, Kulkarni HR, Zhang J, Gerke C, Fimmers R, Kratochwil C, Rathke H, Ilhan H, Maffey-Steffan J, Sathekge M, Kabasakal L, Garcia-Perez FO, Kairemo K, Maharaj M, Paez D, Virgolini I. Prior therapies as prognostic factors of overall survival in metastatic castration-resistant prostate cancer patients treated with [177Lu]Lu-PSMA-617. A WARMTH multicentre study (the 617 trial). Eur J Nucl Med Mol Imaging. 2020 May 8. doi: 10.1007/s00259-020-04797-9.
- Rodrigues M, Winkler KK, Svirydenka H, Nilica B, Uprimny C, Virgolini I. Long-term survival and value of 18F-FDG PET/CT in patients with gastroenteropancreatic neuroendocrine tumors treated with second peptide receptor radionuclide therapy cycle with 177Lu-DOTATATE. (accepted Life)
- Fani M, Weingaertner V, Kolenc Peitl P, Mansi R, Gaonkar RH, Garnuszek P, Mikolajczak R, Novak D, Simoncic U, Hubalewska-Dydejczyk A, Rangger C, Kaeopookum P, Decristoforo C. Selection of the First 99mTc-Labelled Somatostatin Receptor Subtype 2 Antagonist for Clinical Translation-Preclinical Assessment of Two Optimized Candidates. Pharmaceuticals (Basel). 2020 Dec 28;14(1):19. doi: 10.3390/ph14010019.
- Hodolic M, Wu W, Zhao Z, Yu F, Virgolini I, Wang F. Safety and tolerability of 68Ga-NT-20.3, a radiopharmaceutical for targeting neurotensin receptors, in patients with pancreatic ductal adenocarcinoma: the first in-human use. Eur J Nucl Med Mol Imaging. 2020 Oct 2. doi: 10.1007/s00259-020-05045-w. Online ahead of print.
Selection of Funding
- FWF (P30924-B26) Modified Siderophores for “Theranostics” of Aspergillosis 2018 Prof.Dr.Clemens Decristoforo /
- FWF (KLI 909-B) 68Ga-DFO based PET-Imaging of Infections, 2021 Prof.Dr.Clemens Decristoforo/183.965.-
- FWF (I4229-B) Novel 99mTc-labeled somatostatin receptor antagonists 2019 Prof.Dr.Irene Virgolini/230842.-
Patents
Improved pharmacokinetics and cholecystokinin-2 receptor (CCK2R) targeting for diagnosis and therapy (WO/2018/224665)
Collaborations
- Prof. Stefano Fanti, Policlinico S.Orsola-Malpighi, Bologna, Italia
- Prof. Dr. Hans-Jürgen Wester, Technical University Munich, Garching, Germany
- Prof. Dr. Marcus Hacker, Medical University Vienna, Vienna, Austria
- Prof. Dr. Dizdarevic Sabina, Brighton a. Sussex Univ Hospitals NHS Trust (BSUHT), UK
- PD Dr. Ahmadzadehfar Hojjat, Dortmund Klinikum Westfalen, Germany
- Prof. Dr. Mike Sathekge, University of Pretoria and Steve Biko Academic Hospital, SA
- Dr. Milos Petrik, Institute of Molecular and Translational Medicine, Faculty of Medicine and Dentistry, Palacky University
- Prof. Tobias Ross, Department of Nuclear Medicine, Hannover Medical School, Carl-Neuberg-Straße 1, D-30625 Hannover, Germany.
- Dr. Petra Kolenc Peitl, Department of Nuclear Medicine, University Medical Centre Ljubljana, University of Ljubljana, 1000 Ljubljana, Slovenia.
- Prof. Renata Mikolajczak, Radioisotope Centre POLATOM, National Centre for Nuclear Research, 05-400 Otwock, Poland
- Prof. Alicja Hubalewska-Dydejczyk, Department of Endocrinology, Jagiellonian University Medical College, 31-008 Cracow, Poland
- Dr. Jane Sosabowski, Centre for Molecular Oncology, Barts Cancer Institute, Queen Mary University of London
- Dr. Peter Laverman, Radboud University Medical Center Nijmegen, Nijmegen, The Netherlands.
Devices & Services
2 PET/CT, 2 SPECT/CT, Radiopharmacy
 Univ.-Prof. Dr. Irene J. Virgolini
Univ.-Prof. Dr. Irene J. Virgolini
Director
Contact:
Anichstraße 35
6020 Innsbruck
Austria
Email: nuklearmedizin@i-med.ac.at
Phone: +43 512 504 22651
Fax: +43 512 504 22659
http://www.nuklearmedizin-innsbruck.com



